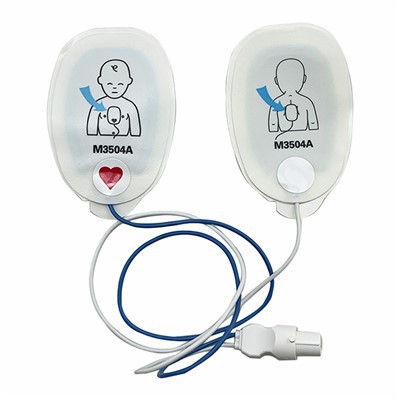
Product Overview and Intended Use:
Our Adult Defibrillator Electrode Patches are high-performance, single-use medical devices designed to be the critical interface between a defibrillator or automated external defibrillator (AED) and a patient experiencing sudden cardiac arrest. These pre-gelled, self-adhesive pads are engineered to deliver life-saving defibrillation shocks and synchronized cardioversion, while simultaneously facilitating ECG monitoring for adult patients. They are essential components in hospital emergency rooms, ambulances, and public access AED programs, where reliability and speed are paramount .
Technical Specifications and Manufacturing Process:
The production of our electrode patches involves advanced material science and precision manufacturing to ensure optimal electrical performance and patient safety.
Conductive Hydrogel Technology: We utilize a proprietary, medical-grade hydrogel formulation. This advanced gel ensures ultra-low impedance for effective energy transfer, maintains superior adhesion to various skin types (including diaphoretic or hairy chests), and is designed to be hypoallergenic to minimize skin irritation. Its unique molecular structure guarantees consistent conductivity and signal clarity for accurate rhythm analysis.
Component Assembly: Each pad consists of multiple layers. The conductive gel is laminated onto a flexible, conformable foam or non-woven backing that allows the pad to adhere firmly to the body's contours. A silver/silver chloride (Ag/AgCl) coated sensor provides a stable electrical pathway. The assembly includes a heavy-duty, reinforced leadwire (available in various lengths, up to a market-leading 60 inches) with a patient connector specifically designed for compatibility with major defibrillator brands (e.g., Physio-Control, ZOLL, Philips, Nihon Kohden) .
Packaging and Sterilization: To preserve gel moisture and ensure sterility, the electrodes are packaged using a high-integrity heat-sealing process in hermetically sealed foil pouches. This packaging, compliant with ISO 11607 standards, guarantees product integrity and a minimum shelf life of 24 to 36 months.
Manufacturer Advantages and Core Competencies:
As a dedicated manufacturer of medical electrical accessories, we offer distinct advantages rooted in decades of specialized experience and vertical integration.
- Vertically Integrated Production: Our facilities handle the entire production process in-house, from hydrogel formulation and coating to die-cutting, printing, and automated packaging. This end-to-end control ensures strict batch-to-batch consistency, rapid prototyping, and the flexibility to accommodate custom OEM/ODM projects, including customized connectors, cable lengths, and multilingual packaging .
- Rigorous Quality Control: Every production batch undergoes stringent in-process and final inspections. We conduct electrical impedance tests, defibrillation recovery tests (to ensure ECG monitoring capability post-shock), and accelerated aging studies to validate performance claims. Our commitment to quality ensures that our electrodes deliver first-shock success when it matters most .
Regulatory Certifications and Compliance:
Our manufacturing quality system and products adhere to the most stringent international regulatory standards, ensuring global market access and patient safety.
- Quality Management System: Our facilities are certified to ISO 13485:2016, the international standard for medical device quality management systems. This certification is maintained and audited by leading global Notified Bodies such as the British Standards Institution (BSI) .
- Global Market Access (MDSAP): We comply with the Medical Device Single Audit Program (MDSAP), which satisfies the regulatory requirements of multiple markets, including Australia, Brazil, Canada, Japan, and the United States, streamlining our ability to serve international clients .
- Product Safety and Performance (CE & FDA): Our defibrillation electrodes are CE marked under the Medical Device Regulation (MDR)
2017/745, demonstrating conformity with European health, safety, and environmental standards. For the U.S. market, our electrodes are FDA-registered and cleared, meeting the rigorous performance requirements of the Federal Drug Administration. Furthermore, all materials are tested for biocompatibility in accordance with ISO 10993 standards, ensuring they are safe for skin contact .
Ordering Information and Customization:
We offer a comprehensive range of electrode pads, including adult and pediatric configurations, for both clinical and training applications. We support low minimum order quantities (MOQs) for pilot programs and scaled production for global distribution. Private label services are available to enhance your brand presence in the market .
Hot Tags: adult defibrillator electrode pads, China adult defibrillator electrode pads manufacturers, suppliers, factory